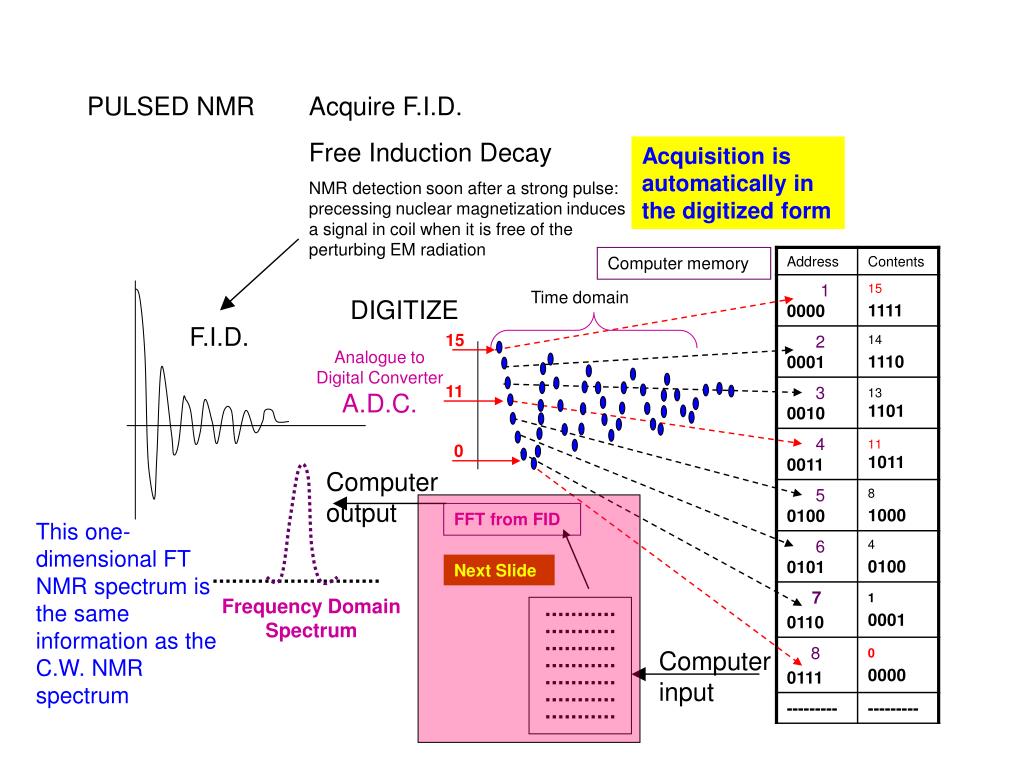
The inherent benefits of giving simultaneous access toīoth qualitative and quantitative evaluation latter isĭefined by the foremost ratio rule: the signal intensity isĭirectly proportional to the number of nuclei that give Quantitative evaluations and is a crucial analytical toolįor the structural determination of unknown syntheticĪs well as natural compounds. NMR has been considered as a rapid,Ĭonvenient, reliable and non-destructive method for Was reported in the year of 1963 by Forbes and Liquids, and its application in quantitative analysis Nuclear Magnetic Resonance (1H NMR) in solids and Purcell andīloch in 1945, discovered the phenomenon of Proton Is an illustrious analytical technique for structureĮlucidation of simple as well as complex moleculesĪnd applied in various branches of bio-sciences,Ĭhemistry, physics and medical diagnosis. Nuclear Magnetic Resonance (NMR) spectroscopy Quantitative nuclear magnetic resonance, validation, internal standard, relative standard Concluding the entire review, various methodologies used for quantification and validations were reflected that proved to be a specific, selective and alternative means of method validation and it can be summarized that nuclear magnetic resonance is as reliable as the chromatography-based technique for quantitative analysis providing accuracy. Studied examples of different concurrent validation methods in the review shows that the results obtained is precise with less standard deviation when compared with other chromatography techniques. In this review numerous examples are cited giving proof of advancement in nuclear magnetic resonance technology and also for precise evaluation of molecular structures of substances in solids and liquid. Validated quantitative nuclear magnetic resonance methods were simple, robust, accurate and selective for evaluating different natural and synthetic samples using various commercially accessible compounds as internal standards.

Hence, this review focuses on the use and feasibility of this technique for quantitative method validation of drug and related compounds.
The quality control and simultaneous quantitation of different residual solvents, analytes and impurities have led analysts to further exploit nuclear magnetic resonance as a viable quantitative technique. Nuclear magnetic resonance spectroscopy has largely given way for most important routine analysis of natural, synthetic drug candidate and related molecules. Höfer, thesis, Universität Stuttgart, 1988. Ernst, Progress in NMR Spectroscopy 16, 163 (1983). Schweiger, in Modern Pulsed and Continuous-Wave Electron Spin Resonance, edited by L. Tsvetkov, Electron Spin Echo Envelope Modulation (ESEEM) Spectroscopy (CRC, Boca Raton, FL, 1992). Wokaun, Principles of NMR in One and Two Dimensions (Clarendon, Oxford, 1987). Mehring, in Modern Pulsed and Continuous-Wave Electron Spin Resonance, edited by L. Lubitz, Electron Nuclear Double Resonance Spectroscopy in Solution (VCH, New York, 1988). Multiple Electron Resonance Spectroscopy, edited by M. Kispert, Electron Spin Double Resonance Spectroscopy (Wiley-Interscience, New York, 1976).
